Reports
COVID-19 and the Family Practitioner: A Message of Hope from a New Outpatient Protocol
This report is based on medical evidence presented at sanctioned medical congress, from peer reviewed literature or opinion provided by a qualified healthcare practitioner. The consumption of the information contained within this report is intended for qualified Canadian healthcare practitioners only.
PHYSICIAN PERSPECTIVE - Viewpoint based on OMA District 11 Doctors' Lounge virtual session - The Semmelweis Reflex on COVID-19 Insights. As well as recently published, peer-reviewed article in American Journal of Therapeutics, April 30, 2021:28:e299-e318
Toronto, Ontario / May 10, 2021
Guest Author:
Pierre Kory, MD, MPA
Pulmonary and Critical Care Specialist
President and Chief Medical Officer of the
Front Line COVID-19 Critical Care Alliance
Former Associate Professor and
Chief of the Critical Care Service at the University of Wisconsin,
Madison, WI, USA
Introduction
Early outpatient treatment is the bedrock of medical care in family practice today. This is as true in the management of COVID-19 as it is for any other potentially progressive condition. Now, with the publication of a comprehensive overview of basic, translational and clinical science supporting a new protocol for the outpatient treatment of early COVID-19, physicians have an authoritative template from which to work that will help minimize the risk of patients progressing to more severe disease, hospitalization and, if not death, then lingering “long-haul” symptoms that can be as debilitating as the acute illness itself. The evidence supporting this new protocol is irrefutable and for the first time, physicians and patients will be able to welcome a message of hope in our battle against this diabolical infection. For more details on the new protocol and how to apply it, Canadian practitioners can turn to the Front Line COVID-19 Critical Care Alliance for on-going support. https://covid19criticalcare.com/
Never in the history of modern medicine have physicians been asked to do so little for so many. Mildly or asymptomatic patients who test positive for the SARS-CoV-2 virus are simply sent home and told to self-quarantine for 14 days. Only if they become short of breath do we open up our hospital doors where patients receive standard-of-care oxygen and, if severely ill, a typically inadequate dose of corticosteroids for too short an interval to make a really significant difference in their outcome. Additional treatments with expensive drugs such as remdesivir are simply no match for COVID-19.
This do-nothing approach is not only unconscionable but it flies in the face of overwhelming evidence in support of therapeutics that can effectively treat outpatient COVID-19 and indeed, prevent infection altogether. In March of 2020, Paul Marik, MD, endowed Professor of medicine, Eastern Virginia Medical School in Norfolk Virginia founded the Front Line COVID-19 Critical Care (FLCCC) Alliance. Together with Marik, Umberto Meduri, MD, Professor of medicine, University of Tennessee Health Sciences Center in Memphis; Joseph Varon, MD, Professor of acute & continuing care, United Memorial Medical Center in Houston, Texas; Jose Iglesias, DO, associate Professor, Hackensack Meridian School of Medicine at Seton Hall, Neptune, New Jersey, and myself, President and Chief Medical Officer of the Front Line COVID-19 Critical Care Alliance, we began to review the current science literature and clinical data to develop a tailored treatment protocol for COVID-19.
At the apex of this evidence is ivermectin, a 40-year-old antiparasitic medication that has been prescribed to roughly 3.7 billion people around the world. Not only have preclinical studies confirmed ivermectin’s activity against SARS-CoV-2 which causes COVID-19, the drug has also been shown to inhibit multiple other viruses in vitro including Zika, Dengue, West Nile and influenza. In a nutshell, ivermectin effectively inhibits replication of the COVID-19 virus. Currently, multiple mechanisms of action have been suggested by studies, the most important one thought to be that ivermectin binds tightly to the coronavirus spike protein, preventing its binding to and entering cells. Further, the multiple, varied mechanisms of action ascribed to ivermectin supports its effectiveness against all COVID-19 variants.
Other studies have identified potent anti-inflammatory properties too, resulting in profound inhibition of multiple inflammatory mediators, the very mediators that can erupt into the well-known cytokine storm to which so many COVID-19 patients succumb prematurely. In a plethora of prevention trials (American Journal of Therapeutics. Published April 30, 2021), people exposed to infected household members were significantly less likely to become sick themselves when given ivermectin immediately. In patients with mild to moderate disease, multiple trials showed that ivermectin consistently hastened recovery and prevented deterioration beyond early symptoms. Even in late treatment trials, ivermectin enabled patients to avoid ICU admission and hastened their recovery; critically ill patients who were clearly headed for death were much less likely to die when treated with ivermectin.
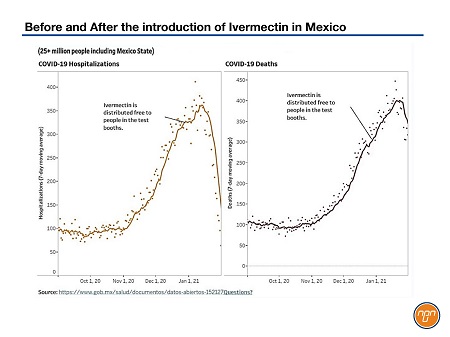
Figure 1.

In short, no other medication has as much evidence supporting its efficacy in all phases of COVID-19 than ivermectin, with protective effects that sometimes equal or even surpass the protection afforded by the vaccines.
It should also go without saying that a drug that has been given to millions of people around the world is safe. In one study involving over 50,000 patients, serious events occurred in less than 1% of the entire cohort (Lancet 1997;350:18-22). Adverse events that do occur were largely due to the body’s inflammatory response to the death of the parasites that the drug was being used to eradicate.
Ivermectin can be given with any medication, however some require close monitoring of dose, the most important being the anticoagulant warfarin and certain immunosuppressants such as the calcineurin inhibitors.
“Test & Treat” Program – Real-World Evidence
Let’s turn to real-world data to see what happened when countries or regions within countries adopted a “Test & Treat” program. Mexico offers up one of the more dramatic examples but it is far from alone.
Back in December of 2020, Mexico was awash in COVID-19. Authorities were desperate, the hospitals were overwhelmed and death rates were sky-high.
So state authorities initiated a “Test & Treat” program where people underwent rapid testing and, if they tested positive, they got ivermectin immediately. Between January 2021 when ivermectin was initially introduced and in March of the same year, hospitalizations and deaths from COVID-19 dropped so precipitously that hospitals have been emptied out and the excess death rate in Mexico is now approaching pre-pandemic levels.
Panama similarly adopted ivermectin nationally in March of this year. Within a few months, both the number of cases and deaths from COVID-19 showed the same steep decline as was seen in Mexico—and it’s not because of some stringent lockdown in Panama either as people were more mobile during the study interval than ever before.
In fact, in every country or at least region within a country where authorities have nationally adopted ivermectin in the treatment of COVID-19—Czech Republic, Slovakia, the states of both Goa and Karnataka in India—the exact same dramatic fall-off of COVID-19 case numbers and deaths is being reported. Even in Delhi, where the program has only been in place since early May of 2021, the curves are heading rapidly downward after what was a seriously awful rise in cases and fatalities from COVID-19 in India starting this March. The world is starting to sit up and notice.
The only agencies which seem immune to the efficacy of ivermectin are the ones who should know better.
I-MASK+ Protocol
Ivermectin is the linchpin for both the treatment of early COVID-19 and its prevention but it does have help from other therapeutics which the FLCCC Alliance has incorporated into its I-MASK+ protocol. Another highly effective medicine included in the protocol is fluvoxamine, supported by research funded by Steven Kirsch, founder of the non-profit, COVID-19 Early Treatment Fund. Fluvoxamine (FLV) has been around almost as long as ivermectin has and again has been prescribed to millions of people around the world.
FLV was originally approved for the treatment of depression and was the first SSRI to be approved for the treatment of obsessive-compulsive disorder in children by the FDA. The evidence supporting its role as an effective treatment of COVID-19 is increasingly powerful. Multiple randomized and observational studies show profound statistically significant reductions in the need for hospitalization when treated with FLV.
Figure 2.
Additionally—and again like ivermectin—the earlier patients are treated, the better the prognosis and clinical outcome. According to evidence provided by www.C19early.com, FLV has the highest effect size of any approved drug for COVID-19. The best example of its efficacy was reported by Seftel et al. in Open Forum Infectious Disease in February, 2021 (https://academic.oup.com/ofid/article/8/2/ofab050/6124100).
In a cohort of COVID-19-positive patients—many of whom were asymptomatic on study enrollment—the sicker ones were actually more likely to take FLV than those who chose observation, biasing results against the FLV group. But it didn’t turn out that way. Zero percent of some 65 patients who opted for FLV required hospitalization compared with 12.5% of those who chose observation alone (P=0.005). Two patients in the observation group required mechanical ventilation, 1 of whom died. At day 14, no patients in the FLV group had any evidence of on-going symptoms compared with 60% of those choosing observation alone (P>0.001).
Even months later, 10% of the observation group still had long-haul COVID-19 symptoms compared with 0% of those treated with FLV. Kirsch is currently conducting a randomized, controlled trial to confirm these results. However, in his experience, any physician who had tried FLV at a dose of 50 mg twice a day for 14 days, initiated as soon as patients knew they were infected, has been “floored” by the drug’s effects.
A third contender in the COVID-19 battle is a seemingly odd, over-the-counter drug, usually thought to be mildly helpful for sleep disturbances—melatonin. As discussed by Richard Neel, MPH, now retired but who worked for the Pentagon for some 20 years where he was tasked with identifying compounds that would protect the military from the harmful effects of biochemical and radiological warfare. This is where, in fact, Neel landed on melatonin as that very compound, whose properties dove-tailed exactly with his need to reduce the harm from potential terrorist destruction.
Neel himself has treated over 2000 patients who developed COVID-19 in the past year, many of whom had significant co-morbidities. Again designed to target early, mildly symptomatic COVID-19 infection, melatonin has also been used as a prophylactic agent as well. In one PLOS Biology study (https://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.3000970), the use of melatonin was associated with an approximately 30% reduction in patients testing positive for SARS-CoV-2 compared with either an ACE inhibitor or an angiotensin II receptor blocker (ARB). Melatonin’s protective effect against COVID-19 infection was even larger among African Americans at about 50%. More information about melatonin can be found at www.lowbluelights.com.
When all is said and done, it would make perfect sense for all physicians to follow the same “Test & Treat” protocol that has resulted in massive successes in multiple countries around the world. Anyone who tests positive for COVID-19 should immediately get ivermectin, at the very least. In addition to the FLCCC Alliance, scientists and physicians around the world have also reviewed the preclinical, clinical and epidemiological evidence surrounding the early outpatient treatment of COVID-19. Many are now advocating for effective protocols for both prevention and early treatment.
The prevention protocols for both high-risk individuals and exposed family members available on the FLCCC Alliance website along with body weight conversions for ivermectin dosing. (www.FLCCC.net).
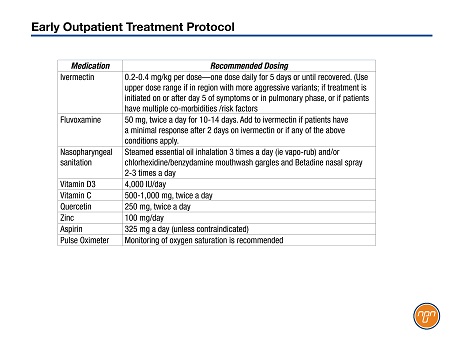
Table 1.
Final Thoughts
With Western countries rolling out vaccine programs at breakneck speed, the question can reasonably be raised: Do we need therapeutics to treat, let alone prevent, COVID-19? The answer is a very loud yes, for a number of reasons. Firstly, unless the virus is eradicated globally, the risk of transmission will continue to lurk in every corner of the world. Secondly, there is an insufficient supply of vaccines to much of the world. Lastly, even if there were, it is not yet clear that they will be effective against the numerous rapidly emerging variants.
Front-line workers are also now witnessing the hospitalization of patients who have already received at least one shot of a COVID-19 vaccine. Clearly, the one-shot-for-everyone-before-two-shots-for-some is not infallible. There is always the reality of vaccine hesitancy as well, which in some countries, especially in some parts of the USA, is widespread. If herd immunity, either by vaccination, or natural immunity acquired in people having already recovered from the illness is not attained, COVID-19 will continue to have devastating effects. So, as long as people continue to get sick, they will need treatment to keep them from progressing to more severe disease, hospitalization and potentially death.
It is time to allow the physicians on the front-line to employ their accumulated training and experience in conjunction with the best available scientific evidence base at the moment they are seeing a sick patient with COVID-19. They know the devastating consequences of untreated disease and can most expertly assess the risk/benefit ratio of treating with these agents vs. not treating. The alternative is following the public health agency approach of waiting the many months to years for unattainably “perfect” evidence before offering treatment to the ill patient seeking care. All focus should instead be placed on ensuring that physicians get the support and freedom to help their patients the best way they know how.
More Info? An ivermectin briefing package including references and peer-reviewed literature is available for medical doctors and health care professionals. It can be found at the following website address: www.mednet.ca You may also request a copy by sending your request to info@mednet.ca We encourage your feedback.
Disclaimer: The opinions expressed in this article are that of the author and do not represent an endorsement by the Ontario Medical Association nor any other professional advisory or regulatory agency, but are endorsed by the Canadian Covid Care Alliance (CCCA) and Front Line Critical Care Alliance (FLCCC) www.flccc.net.
NEW: The Canadian Covid Care Alliance (CCCA), a recently established group of physicians and scientists, welcomes new members signing up for more information at canadiancovidcarealliance.org