Reports
Based on Arthritis Rheum 2012;64(12):3850-5; Arthritis Rheum 2013:65(1):28-38; The Lancet Published online March 18, 2013.
This report is based on medical evidence presented at sanctioned medical congress, from peer reviewed literature or opinion provided by a qualified healthcare practitioner. The consumption of the information contained within this report is intended for qualified Canadian healthcare practitioners only.
MEDI-NEWS - Head-to-Head Comparisons of Biologics in Rheumatoid Arthritis: Closing the Data Gap
March 2013
Over the past decade, the biologics have significantly enhanced the management of patients with rheumatoid arthritis (RA) and continue to do so with the emergence of new biologics with different modes of action. Until recently, no head-to-head comparisons had been done between biologics and selection of treatment for RA was relatively arbitrary given the absence of comparative evidence. However, 3 head-to-head studies have now been published in which the clinical efficacy and safety of different biologics were compared both in short and longer-term trials. While it is not possible to draw hierarchical decisions about which biologic may be superior based on these head-to-head trials, results should help guide treatment selection for patients based on disease severity and individual preference.
Chief Medical Editor: Dr. Léna Coïc, Montréal, Quebec
Monotherapy: Comparing IL-6 with TNF Inhibition
The most recently published head-to-head trial comparing biologics is the ADACTA trial. As approximately one-third of patients with rheumatoid arthritis (RA) receive single agent treatment only, it is important to evaluate the efficacy of biologics in monotherapy. In this study, tocilizumab monotherapy was compared to adalimumab monotherapy (Lancet 2013;Epub ahead of print, March 18).
ADACTA enrolled a total of 326 patients with severe RA at baseline and intolerant to or who could no longer be continued on methotrexate (MTX) therapy. Half of the cohort received tocilizumab 8 mg/kg intravenously every 4 weeks and the other half received adalimumab 40 mg subcutaneously (SC) every 2 weeks.
Importantly, this trial was the first to assess the superiority of one biologic over another and the study showed that response rates were significantly better for all primary and secondary end points.
Unlike adalimumab, a tumour necrosis factor inhibitor (anti-TNF), tocilizumab binds to both membrane-bound and soluble forms of human interleukin-6 (IL-6) receptors, thereby inhibiting signalling mediated by IL-6 and its inflammatory effects.
The primary end point of the study was change in disease activity from baseline to week 24 as assessed by the 28-joint Disease Activity Score (DAS28). When considering clinical efficacy at 24 weeks, it is important to remember that patients in ADACTA had a high level of disease activity (DAS28 >5.1) on study entry. American College of Rheumatology (ACR) 50 and 70 response rates with tocilizumab were significantly higher at 47% and 33% than for adalimumab at 28% and 18% respectively.
As the authors observed, absolute mean swollen and tender joint counts decreased over time in both groups but reductions were more common in the IL-6 receptor inhibitor group. However, the proportion of patients with a swollen joint count of zero at 24 weeks was similar between the 2 groups at 38% for tocilizumab and 30% for adalimumab.
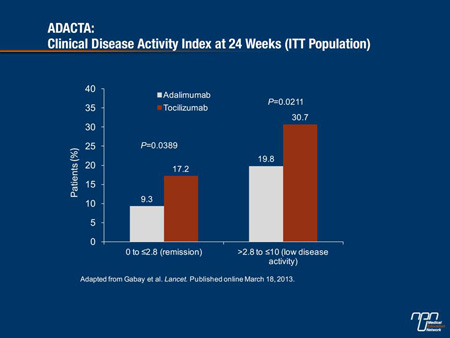
More patients receiving IL-6 receptor inhibitor therapy also achieved Clinical Disease Activity Index (CDAI) remission at week 24 (17.2%) compared to those who received the TNF inhibitor (9.3%; P<0.0389) (Figure 1). From week 16 onwards, more tocilizumab patients also achieved ACR/EULAR remission than adalimumab patients such that at week 24, 18.4% of tocilizumab patients were in ACR/EULAR remission compared with 11.1% of those receiving adalimumab (P=0.0569).
The incidence of adverse events (AEs) was also similar in each group but more patients treated with tocilizumab at approximately 24.7% required dose modification or interruption because of AEs compared with 18.5% of those treated with adalimumab. The most common AEs were infections and infestations, usually mild-to-moderate, serious infections being rare in both groups.
Figure 1.
Comparing an Anti-TNF to a T-cell Modulator
The AMPLE trial (Abatacept versus Adalimumab Comparison in Biologic-Naive RA Subjects with Background Methotrexate) compared the T-cell modulator abatacept 125 mg SC weekly to the anti-TNF adalimumab 40 mg SC biweekly, both in combination with MTX (Arthritis Rheum 2013:65(1):28-38). The mean DAS28-CRP score at baseline was 5.5 and an equal proportion of patients in each group had moderate or high disease activity. A total of 646 patients were randomized to the trial, the majority completing 12 months of treatment.
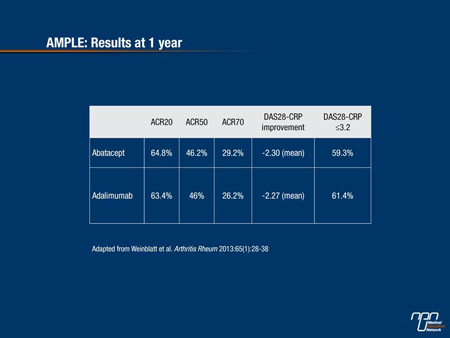
The primary end point was non-inferiority between the 2 regimens as assessed by the ACR 20 response at 1 year. Treatment responses at 1 year of follow-up were similar (Table 1).
Table 1.
As the authors indicated, the proportion of patients in disease remission at 1 year was similar in both treatment groups regardless of which index was used to measure remission. Remission rates according to the ACR/EULAR definition were again similar at 13.5% and 15.7% for the abatacept and the adalimumab treatment groups, respectively.
As the “hallmark” of any substantial RA trial, measures of radiographic damage offer objective and quantifiable outcomes.
In this study, radiographic data showed “similar and robust” improvements in both the erosion and joint space narrowing scores for both groups. Most safety outcomes in the study were comparable between the treatment groups, with similar rates of serious AEs at approximately 10% in each treatment group.
TNF Inhibitor Comparison
Another recently published head-to-head trial compared long-term clinical outcomes with the 2 anti-TNFs, etanercept and adalimumab, with special attention being paid to responses among patients who developed anti-adalimumab antibodies (Arthritis Rheum 2012;64(12):3850-5). The study randomized anti-TNF-naive patients to etanercept 50 mg SC weekly or 25 mg SC twice a week (n=203) or to adalimumab 40 mg SC every other week (n=204). Achievement of clinical outcome was defined as the occurrence of sustained (≥12 consecutive months) low disease activity as reflected by a 28-joint DAS28 <3.2, minimal disease activity (DAS2.8 <2.6) or ACR/EULAR remission based on the Simplified Disease Activity Index (SDAI).
At study entry, patients had active disease (DAS28 ≥3.2) despite earlier treatment with at least 2 disease-modifying anti-rheumatic drugs (DMARDs) including MTX. Fifty-eight percent of adalimumab-treated patients and 55% of their etanercept counterparts completed 3 years of follow-up. Median treatment duration was 143 weeks for the adalimumab group and 130 weeks for those on etanercept. At 3-years follow-up, clinical response rates were similar (Table 2).
Table 2.

Importantly, over one-quarter of adalimumab-treated patients developed anti-adalimumab antibodies during treatment and among these patients, treatment response was considerably poorer: only 13% reached sustained low disease activity although not sustained minimal disease activity, none reached sustained minimal disease activity but not sustained SDAI remission and 4% reached sustained SDAI remission. Corresponding percentages for anti-adalimumab antibody negative patients were 13%, 20% and 20% for the three clinical response categories, respectively. It may be noteworthy that the risk of developing these antibodies was greater among patients who had long-standing severe disease and among those who did not receive concomitant DMARD therapy. As the authors conclude, the overall efficacy of adalimumab and etanercept in RA patients previously naive to anti-TNF therapy is similar.
On the other hand, because such a small proportion of the anti-TNF naive patient group achieved remission in the study, there is a large window of opportunity to increase the efficacy of costly biologic treatments, as the authors suggest.
Summary
Until now, selection of biological DMARDs has been empirical as few comparative studies had been done with biologic agents. For the first time, superiority of tocilizumab over an anti-TNF in monotherapy has been demonstrated, suggesting that interruption of the IL-6 inflammatory pathway is an important contributor to the control of RA. Taken together, results from these comparative trials add to the evidence-base upon which physicians may make informed choices in taking RA disease severity and personal preference into account.