Reports
Hematology in a time of COVID: Update on Paroxysmal Nocturnal Hemoglobinuria (PNH)
This report is based on medical evidence presented at sanctioned medical congress, from peer reviewed literature or opinion provided by a qualified healthcare practitioner. The consumption of the information contained within this report is intended for qualified Canadian healthcare practitioners only.
PRIORITY PRESS - EHA2021 Virtual – European Hematology Association (EHA)
Online / June 9–17, 2021
Online – This year’s European Hematology Association meeting (EHA2021) covered the new medical realities of hematology during the COVID-19 pandemic. Expert panels discussed the impact of SARS-CoV-2 on clinical decision-making in prothrombotic hematological diseases such as PNH. Despite the rarity of the disease, data and commentary on PNH were prominent in the five-day meeting due to rapid advances in the field. In addition to discussing COVID-19 vaccination in patients with PNH, presenters updated delegates on new therapies aiming to join gold-standard eculizumab in clinical practice. Nearly 20 PNH abstracts and e-posters and 12 scientific presentations featured studies on pegcetacoplan, ravulizumab and first-in-class oral therapy iptacopan, among others.
Chief Medical Editor: Dr. Léna Coïc, Montréal, Quebec
“The most vicious acquired thrombophilic state in medicine,” said Dr. Austin Kulasekararaj about PNH, quoting pioneer Prof. Lucio Luzzatto. PNH is a rare chronic and life-threatening disease of uncontrolled terminal complement activation, leading to intravascular hemolysis, thrombosis and end-organ damage. Dr. Kulasekararaj leads the King’s College Hospital PNH centre, one of the U.K.’s two PNH hubs.
During a talk at EHA2021, Dr. Kulasekararaj reminded his audience that prior to the availability of C5-inhibitor eculizumab 35% of patients, most in their early 30s, died within 5 years of diagnosis despite best supportive care. However, while the prognosis has improved, diagnosis remains a challenge, he said.
“It’s All in Your Head”: Challenges of PNH Diagnosis
PNH diagnosis can take 10 years or more – assuming the patient survives that long, said Dr. Kulasekararaj. He presented the case of a 39-year-old female patient with supposed hematuria, chronic fatigue and, according to the presenter, a “pretty decent” hemoglobin of 110–120 g/L. “When I saw her she was already on a couple of antidepressants and she was branded as thinking that everything [was] psychological,” Dr. Kulasekararaj explained. Three years after the initial presentation, hemoglobinuria, dysphagia and abdominal pain developed and testing revealed
a red-cell PNH clone of 49.2%.
In an expert panel, Dr. Wolfgang Füreder of the Medical University of Vienna warned, “In PNH, the blood [count] can be completely normal – one has to consider this.” Audience member Hadeel Ammar asked whether all patients with normal hemoglobin and unprovoked thrombosis to an unusual site should thus be investigated for PNH. Dr. Füreder said that elevated lactate dehydrogenase (LDH) pointed towards PNH.
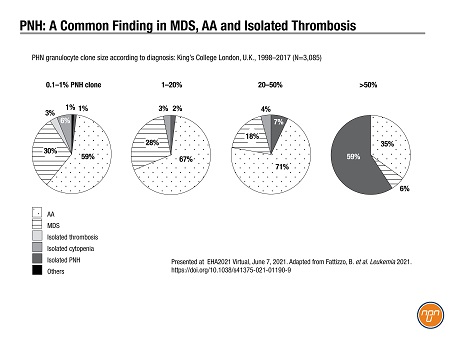
PNH diagnosis is particularly troublesome in other hematological settings. At the conference, Dr. Kulasekararaj shared 19 years of data from King’s College London1 showing that most patients with a PNH clone of <50% had diagnoses of either MDS, aplastic anemia or isolated thrombosis (N=3,085; see Figure 1).
Figure 1.
COVID-19 and Other Infections
Speakers shared their experience with COVID-19 vaccination in patients with PNH. Dr. Robert Brodsky, Director of the Division of Hematology at Johns Hopkins University, said one-third to one-half of his patients experienced breakthrough hemolysis requiring transfusion after the vaccine resulted in a complement-amplifying condition.
Patients and physicians should be warned that such reactions “aren’t unusual” and resolve within a few days. Dr. Brodsky concluded: “The risk-benefit [of the vaccine] is in favour of the patient.”
Although there are no data on COVID-19 infection in PNH yet, complement-associated defects in PNH are known to predispose patients to infection – mostly bacterial. An EHA poster led by Dr. Bhumika Patel of the Taussig Cancer Institute, Cleveland Clinic, attempted to quantify the scale of the risk. Patel et al. compared incidence rate of serious infections requiring hospitalization in 1,083 patients with PNH and 3,249 control patients using the IBM MarketScan Commercial Database.2 Serious infection, mostly bacterial, was eight times as likely in the PNH patients (incidence/100 person-years: 12.7 versus 1.7, PNH and control, respectively).
Dr. Louise Arnold of the U.K.’s Leeds PNH centre presented “the largest experience” of successful management of infection risk due to Neisseria meningitidis in PNH patients treated with complement inhibition.3 Between 2002 and 2020, 324 U.K. patients received 801 MenACWY vaccination; from 2015, 287 patients also received Bexsero (MenB vaccine). Over 18 years, the centre had nine cases of meningococcal sepsis, with one death, likely due to delay in seeking treatment. The authors concluded, “it is essential patients are vigilant for fever, seeking immediate medical attention stating their diagnosis of PNH on complement inhibitor therapy”.
New Therapeutic Options
Speakers during EHA2021 referred to the “exciting” prospect of several new therapies for PNH.
Current standard eculizumab, a C5-inhibitor, is now joined by derivative ravulizumab in the European Union and the U.S. (Ravulizumab is not currently available in Canada.) Ravulizumab offers a longer half-life and less frequent dosing than eculizumab. Dr. Srikanth Nagalla, Miami Cancer Institute, presented real-world U.S. data at EHA2021 comparing eculizumab and early data for ravulizumab (N=1,330).4 In an email interview Dr. Nagalla cautioned that the ravulizumab sample size was small; however, he and his colleagues noted “Similar trends… between treatment groups.”
Proximal complement inhibitors that featured at EHA2021 included C3-inhibitor pegcetacoplan, which is administered by s.c. infusion in the abdomen every three days. Posters led by Dr. Regis Peffault de Latour, Université de Paris, and
Dr. Alexander Röth, University of Duisberg-Essen, on the 48-week extension of the PEGASUS registration trial showed that 73% of the patients on pegcetacoplan remained transfusion-free at 48 weeks,5 with improvements in fatigue and dyspnea close to population norms6. The trial was in eculizumab
non-responders.
In PEGASUS, pegcetacoplan significantly improved mean Hb level versus eculizumab by Week 16 (Peg, n=41; Ecu, n=39);5 85% of the pegcetacoplan patients avoided transfusion versus 15% of the eculizumab patients. During a review of new therapies Dr. Brodsky said that these results were not unexpected in view of the fact that 80% of the eculizumab patients were transfusion-dependent at baseline.
The most common adverse events (AEs) for pegcetacoplan in PEGASUS were injection-site reactions (36%), hemolysis (24%) and diarrhea (21%); 30% experienced serious AEs, 6% of which were “possibly drug-related”.5 Dr. Brodsky presented data on breakthrough hemolysis on the two drugs and noted that high levels of LDH were seen in the pegcetacoplan patients not seen with eculizumab. “These are high LDHs that worry us about the potential increased risk of thrombosis,” Dr. Brodsky said. “This needs further looking into.” (Pegcetacoplan is not approved in Canada.)
First-in-class PNH oral therapies at the conference included iptacopan (LNP023), a factor B inhibitor. Dr. Jun Ho Jang from Sungkyunkwan University in Seoul presented results of a phase-2, dose-finding study of iptacopan in C5-inhibitor-naïve patients. Doses of iptacopan up to 200 mg lowered LDH by at least 60% in all patients (N=13).7 All but one of the patients remained transfusion-free up to week 12. No serious AEs were reported. Session chair Dr. Kulasekararaj raised theoretical concerns about increased infections with proximal inhibition; Dr. Ho Jang said there was no evidence of this so far, “but we need more data.”
Conclusions
PNH is a rare, progressive, life-threatening disease whose diagnosis is often obscured by the presence of other hematological disorders. Delegates were urged to check LDH levels in the presence of anemia and unexplained thrombosis. COVID-19 vaccination offers a favourable risk-benefit for patients but physicians should be aware of the potential for transient breakthrough hemolysis. Patients with PNH should seek immediate medical attention for any fever and make ER staff aware of their status. New therapies for PNH include inhibitors of C3 and factor B in the proximal complement pathway.
References:
1. Fattizzo B, et al. Clinical and prognostic significance of small paroxysmal nocturnal hemoglobinuria clones in myelodysplastic syndrome and aplastic anemia. Leukemia. Published online March 4, 2021. https://doi.org/10.1038/s41375-021-01190-9
2. Patel BJ, et al. Risk of serious infections in patients with or without paroxysmal nocturnal hemoglobinuria: A real-world matched cohort analysis. EHA2021 Virtual June 9–17, 2021. Poster EP596.
3. Arnold L, et al. Management of meningococcal disease risk in patients with paroxysmal nocturnal hemoglobinuria (PNH) on complement inhibitors: 18 years’ experience from the UK National PNH Service in Leeds. EHA2021 Virtual June 9–17, 2021. Poster EP592.
4. Nagalla S, et al. Ravulizumab and eculizumab reduce transfusions in adult patients with paroxysmal nocturnal hemoglobinuria: Evidence from three real-world databases: TriNetX U.S. EMR, TriNetX U.S. Claims and KOMODO Health. EHA2021 Virtual June 9–17, 2021. Poster EP1337.
5. Peffault de Latour R, et al. Forty-eight week efficacy and safety of pegcetacoplan in adult patients with paroxysmal nocturnal hemoglobinuria and suboptimal response to prior eculizumab treatment. EHA2021 Virtual June 9–17, 2021. Poster S174.
6. Röth A, et al. Effect of pegcetacoplan on quality of life in patients with paroxysmal nocturnal hemoglobinuria: week 48 of PEGASUS phase 3 trial comparing pegcetacoplan to eculizumab. EHA2021 Virtual June 9–17, 2021. Poster EP595.
7. Ho Jang J, et al. First-line treatment of PNH patients with iptacopan leads to rapid and durable hemoglobin increase by controlling both intra- and extravascular hemolysis. EHA2021 Virtual June 9–17, 2021. Poster S173.