Reports
Regina Qu’Appelle Health Region
This report is based on medical evidence presented at sanctioned medical congress, from peer reviewed literature or opinion provided by a qualified healthcare practitioner. The consumption of the information contained within this report is intended for qualified Canadian healthcare practitioners only.
PHYSICIAN PERSPECTIVE - Get Up to Date - A Regional Perspective on the Evidence and Resulting Changes in ACS Protocol
May 2013
Reviewed and edited by:
William Semchuk, MSc, PharmD, FCSHP
Manager, Clinical Pharmacy Services
Regina Qu’Appelle Health Region
Assistant Clinical Professor
College of Medicine, and College of
Pharmacy and Nutrition
University of Saskatchewan,
Regina, Saskatchewan
Introduction
Antiplatelet therapy options in the management of acute coronary syndromes (ACS) have recently been updated within the Regina Qu’Appelle Health Region (RQHR). Recent changes in the RQHR formulary provide the opportunity to significantly reduce the risk of major cardiovascular (CV) events by employing more effective antiplatelet strategies compared to the previous standard of clopidogrel and ASA. The most significant change, based on a large trial, is the recommendation to employ ticagrelor to provide additional protection against major CV events in certain patient groups. Guided by the large clinical trials that underlie the change in formulary, the new recommendations allow for the clinical gains from greater antiplatelet effect, including in some cases, a lower risk of death. They also balance benefit with an acceptable low risk of major or minor bleeding. Due to the fundamental importance of deactivating platelets to alter the natural history of evolving ACS events, optimal use of antiplatelet therapy should be considered an essential strategy for improving the prognosis in ACS.
Previous Standard: Opportunity for Improvement
The dual antiplatelet strategy of clopidogrel and ASA in patients presenting with ACS has been a widely employed standard for more than 10 years. Despite the use of ASA and clopidogrel, rates of CV events in ACS populations remain substantial. In the landmark CURE study, 10% of those receiving clopidogrel plus ASA went on to suffer a recurrent myocardial infarction (MI), a persistent arterial occlusion or died of a CV cause despite the 20% reduction in these risks with the combination relative to ASA alone.1 This study was performed in patients with non-ST elevation MI (NSTEMI). In the CLARITY-TIMI 28 trial, conducted in patients with ST elevation MI (STEMI), the residual risk of death, stroke or MI was 9% in the group receiving clopidogrel plus ASA despite a 31% reduction in risk of a major CV event relative to ASA alone.2
Since those studies established this dual antiplatelet combination as a standard in ACS, two large ACS trials have proven that more effective antiplatelet therapy will further reduce CV risk. One trial tested ticagrelor in an all-comer population of ACS patients.3 The other study tested prasugrel in ACS patients scheduled for a percutaneous coronary intervention (PCI).4 Data from these trials provide an opportunity to improve outcomes over the previous clopidogrel plus aspirin standard.
In the TRITON TIMI-38 study, 13,608 ACS patients scheduled for PCI were randomized. In the experimental arm, patients received a loading dose of prasugrel (60 mg) followed by maintenance prasugrel (10 mg daily). The comparator arm received a loading dose of clopidogrel (300 mg) followed by maintenance clopidogrel (75 mg daily). Both groups received ASA. Approximately 25% of the ACS events were STEMI and the remaining NSTEMI.
Relative to clopidogrel, prasugrel reduced the risk of the composite end point of death from CV cause, MI or stroke by 19% (HR 0.81; P<0.001). The risk of major bleeding on prasugrel was increased by 32% (HR 1.32; P=0.03) relative to clopidogrel. There was no difference in mortality. The authors concluded that the greater protection against ischemic events must be weighed against an increased risk of bleeding, but post-hoc analyses provided guidance for candidate selection.
In the PLATO trial, individuals admitted to hospital with ACS were randomized regardless of planned procedure or pre-hospital antiplatelet treatment. The experimental arm received a loading dose of ticagrelor (180 mg) followed by maintenance ticagrelor (90 mg twice daily). The comparator arm received a loading dose of clopidogrel (300 or 600 mg) followed by maintenance clopidogrel (75 mg daily). Both groups received ASA. Approximately 37% of the 18,624 patients randomized had STEMI and the remaining had NSTEMI.
Relative to clopidogrel, ticagrelor reduced the risk of the composite end point of death from vascular causes, MI or stroke by 16% (HR 0.84; P<0.001). The difference in total major bleeding (11.6% vs. 11.2%; P=0.43) did not reach statistical significance but non-CABG major bleeding was significantly increased (4.5% vs. 3.8%; P=0.026). Noteworthy to antiplatelet trials, ticagrelor was associated with a 16% reduction (HR 0.84; nominal P<0.001) in all-cause mortality.
New Data Translated into Clinical Practice
Findings from both TRITON-TIMI 38 and PLATO trials resulted in discussions pertaining to the addition of other antiplatelet options to the RQHR formulary. In order to streamline the treatment process, the decision was made to only add one of the two new agents to formulary.
The agent selected for formulary inclusion within the RQHR was ticagrelor. It provides clinicians the opportunity to improve outcomes based on the PLATO trial. While all ACS patients should be initiated on ASA immediately, the second antiplatelet agent is defined by the diagnosis, the planned strategies for intervention and specific patient characteristics. Several large organizations have altered ACS antiplatelet guidelines on the basis of the PLATO and TRITON-TIMI 38 trials, but specific treatment approaches at the regional or hospital level are appropriate because of differences in ACS care.
At regional tertiary care centres, including the Regina General Hospital, the data generated by the PLATO and TRITON-TIMI 38 trials can be applied directly. In both STEMI and NSTEMI patients, ticagrelor is now one of the options in a dual antiplatelet strategy with ASA, however clopidogrel remains the most commonly used antiplatelet agent in combination with ASA at the Regina General Hospital. Currently, ticagrelor is predominantly used in combination with ASA 75 mg to 150 mg daily in situations in which the Saskatchewan Prescription Drug Plan has provided coverage. Namely in those patients with ACS, i.e., STEMI, NSTEMI or unstable angina (UA) according to certain criteria (Table 1).
Table 1. Antiplatelet Use Recommendations at Regina Qu’Appelle Health Region

In STEMI patients scheduled for PCI, prasugrel has been found more effective than clopidogrel in a dual antiplatelet strategy with ASA, but the benefit-to-risk ratio varied across subpopulations in the registration trial, producing some important exceptions. As a result, clopidogrel not only remains the preferred partner with ASA in individuals who have received anticoagulants or prior treatment with a fibrinolytic agent but in those over the age of 75 years, those who weigh <60 kg and those with a prior transient ischemic attack or stroke. In these individuals, the evidence suggests that the greater risk of major bleeding negates the clinical benefit prasugrel.
Relevance of New Options to Regional Centres
The objective evidence that newer antiplatelet therapies can improve outcomes in ACS patients informs but does not dictate adjustments in patient care. Due to substantial regional disparities in the care of ACS, driven largely by variability in resources such as the proximity to catheterization laboratories and differences in transfer intervals to catheterization laboratories, treatment guidelines must be adjusted for relevance to current practice.
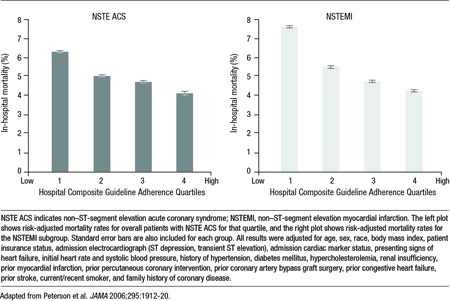
There are compelling data to conclude that implementation of more modern strategies for appropriate candidates will improve ACS outcomes including a reduction in mortality. The implementation and adherence to treatment guidelines in the management of ACS has been associated with statistically significant improvements in outcome. In an observational analysis that included 350 academic and non-academic centers, a stepwise 10% reduction in in-hospital mortality rates was associated with each 10% increase in adherence to evidence-based guidelines (Figure 1).5
Figure 1. Association Between Hospital Composite Guideline Adherence Rate and In-hospital Mortality
Conclusion
The first-line antiplatelet strategies in ACS patients have been revised. The newer agent ticagrelor provides an important opportunity to improve outcomes relative to clopidogrel when it is combined with ASA. In NSTEMI patients, the advantage of ticagrelor over clopidogrel in appropriately selected patients includes a mortality reduction.
Question & Answers
Q: What is your perspective on the benefit-to-risk ratio that the newer antiplatelet agents offer within the revised options for reducing the risk of thrombosis within an acceptable rate of bleeding?
A: The PLATO trial demonstrated that ticagrelor as compared to clopidogrel decreased the primary end point (a composite of death from vascular causes, MI or stroke) from 11.7% to 9.8% (HR, 0.84; 95% CI, 0.77-0.92; P<0.001). The rate of death from any cause was also reduced with ticagrelor (4.5%, vs. 5.9% with clopidogrel; P<0.001). Within the PLATO trial there was no significant difference in the rates of major bleeding found between the ticagrelor and clopidogrel groups (11.6% and 11.2%, respectively; P=0.43), however ticagrelor was associated with a higher rate of major bleeding not related to CABG (4.5% vs. 3.8%, P=0.03), including more instances of fatal intracranial bleeding and fewer of fatal bleeding of other types. Keeping appropriate patient selection in mind, these bleeding events must be kept in the context of overall mortality reduction.
Q: The studies that led to changes in the options compared therapies in different populations. What insights can you offer on why it was important to prove superiority of prasugrel or ticagrelor over clopidogrel in different ACS groups (STEMI, NSTEMI, unstable angina, etc)?
A: The demonstration of superiority over standard therapy is important as clinicians must always weigh the benefit of the new therapy against adverse effects, cost and the limited information pertaining to use outside of a clinical trial setting. By demonstrating benefit in a broad array of ACS patients (STEMI, NSTEMI, unstable angina and those going onto coronary artery bypass) clinicians can be comforted by these findings in a broad array of ACS patients.
Q: What is your point of view on the possible side effects associated with the newer agents vs. the opportunity to improve outcomes?
A: As new agents are brought into the therapeutic armamentarium, clinicians must always be aware of the potential emergence of untoward adverse events. In this large clinical trial of over 18,000 patients, dyspnea occurred more frequently in patients receiving ticagrelor than clopidogrel (13.8% vs. 7.8%) and dyspnea resulting in drug discontinuation occurred more commonly as well (0.9% vs. 0.1%). Ventricular pauses occurred more frequently in patients receiving ticagrelor as compared to clopidogrel as well. It will be important to continue to document adverse effects of ticagrelor (like any new drug) to ensure that a representation of adverse effects is characterized in a real world setting.
Q: The options introduce some decision points not previously required when all patients were treated with clopidogrel plus ASA. What action needs to be taken to improve outcomes?
A: At the present, the clinician must weigh multiple variables including clinical trial evidence, patient characteristics and presentation, medication adverse effect profile, availability and coverage of new agents along with patient’s willingness to pay for agents if not covered. By being aware of the clinical trial evidence within the context of a patient scenario and by involving the patient in the decision of whether cost is a prohibitive factor, the physician will be able to customize the therapeutic approach to the individual.
References
1. Yusuf et al. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N Engl J Med 2001;345(7):494-502.
2. Sabatine et al. Addition of clopidogrel to aspirin and fibrinolytic therapy for myocardial infarction with ST-segment elevation. N Engl J Med 2005;352(12):1179-89.
3. Wallentin et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2009;361(11):1045-57.
4. Wiviott et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2007;357(20):2001-15.
5. Peterson et al. Association between hospital process performance and outcomes among patients with acute coronary syndromes. JAMA 2006;295(16):1912-20.