Reports
Type 2 Diabetes and Antihyperglycemics: A View from the Kidney
This report is based on medical evidence presented at sanctioned medical congress, from peer reviewed literature or opinion provided by a qualified healthcare practitioner. The consumption of the information contained within this report is intended for qualified Canadian healthcare practitioners only.
PRIORITY PRESS - Annual General Meeting of the Canadian Society of Nephrology
Montreal, Québec / April 24-28, 2013
Montreal - A new class of antihyperglycemic agents, the sodium-glucose co-transporter 2 inhibitors significantly reduce both blood pressure and weight beyond achieving glucose-lowering effects through an insulin-independent mechanism. As such, there is virtually no risk of inducing hypoglycemia when used alone. For patients with type 2 diabetes and chronic kidney disease (CKD), several agents other than insulin may now be used to lower hemoglobin A1c to target levels, but not all classes may be used freely in CKD and even drugs within the same class of hypoglycemic agents may differ in terms of their suitability for glycemic control.
Chief Medical Editor: Dr. Léna Coïc, Montréal, Quebec
“The kidney plays a major role in glucose homeostasis not only because it reabsorbs glucose but it actually produces glucose through gluconeogenesis,” Dr. Jordan Weinstein, Assistant Professor of Medicine, University of Toronto, Ontario told delegates here at the congress. In healthy people, the kidney reabsorbs precisely the same amount of glucose as is filtered out of the blood through the kidney, resulting in a zero sum gain, he added. Ninety per cent of the glucose is reabsorbed in the kidney by the sodium-glucose co-transporter 2 (SGLT2), the other 10% being reabsorbed through the SGLT1. “If all is working well, the 2 transporters are not overwhelmed and no glucose is excreted in the urine,” Dr. Weinstein observed.
However, as a way to control type 2 diabetes, the use of SGLT2 inhibitors is very appealing as it prevents glucose to be reabsorbed by the kidney and can cause a net caloric loss. Notably, sodium is not reabsorbed either, meaning that SGLT2 inhibitors also act as a mild diuretic. Thus, unlike most antihyperglycemic agents in use today, SGLT2 inhibitors predictably cause weight loss. “And because these inhibitors only inhibit reabsorption of glucose and have no effect on insulin, there’s little risk for hypoglycemia which is a very attractive feature,” Dr. Weinstein added.
Glucose, Weight and Blood Pressure
Dr. Catherine Yu, Assistant Professor of Medicine, University of Toronto, reviewed clinical trial data of the hypoglycemic effects of canagliflozin and dapagliflozin used alone and as add-on agents. Summarizing results seen with canagliflozin, “generally, the A1c reduction is between 0.6% and 0.9% in each of these studies, depending on the dose, which is about the same as metformin,” she observed.
Across the same studies, “there is a dose-dependent reduction in weight ranging from 2 kg to 5 kg which is comparable to what we see with the glucagon-like peptide-1 (GLP-1) receptor agonists,” she added. The reductions of approximately 3 to 5 mmHg in systolic blood pressure consistently seen with the SGLT2 inhibitors are also clinically meaningful. When canagliflozin is added to metformin, the incidence of hypoglycemia is low at around 5% but it is higher when added to a sulfonylurea (SU) because SUs induce hypoglycemia, as Dr. Yu also pointed out.
SGLT2 Inhibition in Moderate Renal Impairment
Because of their mechanism of action, it is reasonable to ask how well the SGLT2 inhibitors perform in those with chronic kidney disease (CKD). In a recent study by Yale et al., 269 patients were randomized to canagliflozin 100 or 300 mg a day or placebo (Diabetes Obes Metab 2013;15(5):463-73). All patients were in stage 3 CKD with an estimated glomerular filtration rate (eGFR) of ≥30 and <50 ml/min/1.73 m2. At week 26, both doses of the SGLT2 inhibitor reduced A1c from baseline by 0.33% and 0.44% for the 100 and 300 mg doses, respectively, compared with a 0.03% reduction with placebo (P<0.05).
Overall adverse event rates were similar for both doses of canagliflozin and placebo although slightly higher rates of urinary tract infections were observed with the higher dose of active therapy. These findings indicate that canagliflozin remains reasonably effective in moderate renal impairment, as Dr. Yu suggested.
In Europe, dapagliflozin has been licensed for use in type 2 diabetes but it is not licensed for use in the setting of moderate renal impairment, as Dr. Yu also noted. In the US where canagliflozin is licensed, physicians may use the 100 mg dose for patients with an eGFR of 45 to 60 ml/min/1.73 m2 and the 300 mg dose for those with higher levels of renal function.
The effect of causing glycosuria through SGLT2 inhibition does increase the risk for genital mycotic infections, especially in females. However, experience suggests these infections tend to be solitary yeast infections and do not typically recur. “We need to make patients aware of this increased risk,” Dr. Sheldon Tobe, Professor of Medicine, University of Toronto cautioned. “But if people are aware of the risk and we identify and treat these infections early, there should not be a major concern.”
End-Stage Renal Disease and Hypoglycemic Agents
Fewer than 10% of patients with type 2 diabetes go on to develop end-stage renal disease (ESRD) and require dialysis. Importantly, because so many patients have type 2 diabetes worldwide, that small proportion translates into over half of type 2 diabetes patients in dialysis units. For these patients, it is important to know which hypoglycemic agents can be used at which stage of CKD. “With the new Canadian guidelines, we have tended to personalize A1c targets a bit more by allowing the goal to be as low as 6% for mostly early diabetes because if we control diabetes early on, we can reduce microvascular complications,” Dr. Jean-Francois Yale, Professor of Endocrinology, McGill University, Montreal, Quebec, told delegates. “As long as we can use agents that do not cause hypoglycemia, we can aim for perfect (<7%) control,” he added. Once forced to use medications that cause hypoglycemia, “7% will become a more reasonable goal and 8% is also reasonable for those with limited life expectancy or where there are only disadvantages if we are too aggressive.”
As in previous guidelines, the new diabetes guidelines (www.guidelines.diabetes.ca) recommend physicians start with metformin; when metformin is not sufficient to achieve target A1c goals, “we must individualize,” Dr. Yale stated. Part of deciding which agent to add depends on the patient’s kidney function: here, hypoglycemic drug classes vary as to their suitability for use in CKD and there is some difference even between drugs in the same class.
Broadly speaking, metformin may be used at full dose in patients with a GFR >45 mL/min/1.73 m2 and reduced by half in those with a GFR <45 mL/min/1.73 m2 . Use of the SUs in CKD patients varies. Caution should be exercised when using glyburide in patients with a GFR <50 mL/min/1.73 m2 and stopped when it is <30 mL/min/1.73 m2 . The other SUs appear to be safer in CKD and gliclazide can be used down to a GFR of 15 mL/min/1.73 m2 , while repaglinide can be used at any level of kidney function including in dialysis patients.
Figure 1. Antihyperglycemic Agents and Renal Failure
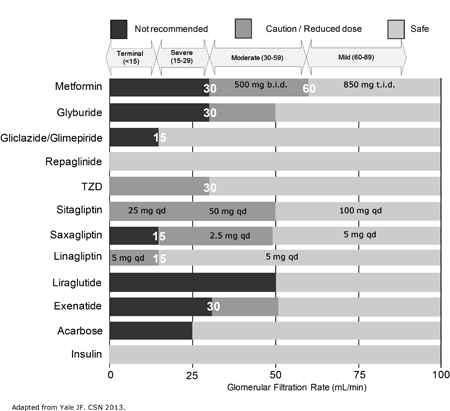
The thiazolidinediones are not widely used because of their side effect profile, as Dr. Yale pointed out; if they are used in CKD patients, they should be prescribed carefully in patients with a GFR <30 mL/min/1.73 m2 .
As for the DPP-4 inhibitors, sitagliptin does accumulate with diminishing renal function. To counteract this, it is recommended that physicians reduce the standard dose to 50 mg for patients with a GFR between 30 and 50 mL/min/1.73 m2 and to 25 mg for those with a GRF <30 mL/min/1.73 m2. Saxagliptin is not indicated in patients with a GFR <15 mL/min/1.73 m2 but it can be used at a reduced dose of 2.5 mg in those with a GFR of 15 to <60 mL/min/1.73 m2. Linagliptin is indicated in patients with a GFR <15 mL/min/1.73 m2, with no dose adjustment. Few to no studies have been done with the injectable GLP-1 receptor agonists liraglutide and exenatide in renal failure so neither is approved for use in GFR <50 mL/min/1.73 m2.
Finally, the SGLT2 inhibitors do vary in terms of their recommended use in CKD patients, as per their licensing indications. “Insulin of course can be used at any level of renal failure but we are bit more fortunate than we were even 2 years ago as we now have patients on dialysis in whom we can use several other agents other than just insulin,” Dr. Yale concluded.
Summary
The new Canadian Diabetes Guidelines, released in April, 2013, still recommend metformin as first-line therapy for patients with type 2 diabetes. After that, physicians must individualize according to duration of disease, the presence of comorbidities and life-expectancy. Among the comorbidities that make treatment of type 2 diabetes more challenging is CKD, as not all hypoglycemic drug classes are indicated in patients with failing renal function and not even all drugs within the same class have the same recommendation. It is therefore important to appreciate which drugs may be used within specific levels of CKD for patients with type 2 diabetes and follow licensing indications carefully.